Leucine Production Technology
Production process advantages: Microbial fermentation method
Process research and optimization services: Provide formula development, process design, pilot-scale to production-scale trials, and parameter optimization to enhance efficiency and reduce costs
Production technical support: Provide equipment commissioning, operation training, and on-site guidance to ensure stable production operation
Technical consultation and solution services: Provide overall technical services such as project planning, production line design, and process schemes
After-sales operation and maintenance services: Provide equipment maintenance, fault handling, and process upgrading for long-term technical support
Process scale-up and industrialization services: Provide pilot-scale, production-scale, process scale-up, and industrialization implementation services
The foundation for successful application of production technology: 30% process + 70% local execution
- Fast Delievery
- Quality Assurance
- 24/7 Customer Service
Product Introduction
Leucine Production Technology Solution Provider
Shaanxi Zhenhuacui Biotechnology delivers cutting-edge Leucine Production Technology through progressed microbial maturation strategies. Our enterprise-grade arrangement utilizes metabolically designed Corynebacterium glutamicum strains to accomplish leucine yields surpassing industry benchmarks. With over 10 a long time of generation encounter and 8 a long time in wellbeing nourishment investigate, we give total turnkey frameworks from pilot-scale to commercial generation. Our innovation guarantees 91% downstream feed-grade applications whereas assembly pharmaceutical-grade virtue prerequisites. Standard shipments available within 20 days, customized configurations take anywhere from 30 to 90 days, depending on the specific specifications.
Technology Overview
Our item speaks to the apex of present day biotechnology fabricating. We've aced the complex microbial maturation prepare that changes basic carbon sources into high-purity leucine through built bacterial metabolism.
The innovation centers on optimized Corynebacterium glutamicum strains that have been carefully altered to overcome normal input hindrance. These strains effectively change over glucose and corn-based substrates into leucine whereas minimizing undesirable by-products like valine and isoleucine.
Our framework coordinating progressed handle control with real-time observing. Temperature control between 31-37°C and exact pH control at 6.4-7.4 guarantees ideal protein action. Energetic oxygen administration bolsters oxygen consuming digestion system whereas keeping up perfect aging conditions.
The breakthrough lies in our restrictive strain advancement. We've upgraded the branched-chain amino corrosive amalgamation pathway, coordinating metabolic flux particularly toward leucine generation. This focused on approach conveys predominant yields compared to ordinary methods.

Production Process
Upstream Preparation
Our prepare starts with thorough strain development and medium planning. High-performance bacterial strains experience multi-stage seed development to accomplish ideal cell thickness and metabolic activity.
Core Fermentation
The heart of our Leucine Production Technology involves precisely controlled fermentation in sterile bioreactors. Our automated systems monitor:
- Bacterial concentration and viability
- Product aggregation rates
- Nutrient utilization patterns
- Environmental parameters
Downstream Processing
Advanced partition innovations extricate and filter leucine from complex aging broths. Our multi-step decontamination handle incorporates filtration, centrifugation, and crystallization to accomplish target virtue levels.
Quality Control
Every clump experiences comprehensive testing for immaculateness, microbial limits, and overwhelming metal substance. Items meet worldwide benchmarks for nourish, nourishment, and pharmaceutical applications.
Technical Advantages
Superior Productivity: Our metabolically optimized strains convey higher leucine yields than conventional aging methods.
Scalable Plan: From 50L pilot frameworks to 500,000L commercial generation, our innovation scales seamlessly.
Environmental Supportability: Green maturation forms minimize squander era and vitality utilization compared to chemical synthesis.
Flexible Generation: Single stage produces feed-grade, food-grade, and pharmaceutical-grade leucine by altering refinement parameters.
Proven Unwavering quality: 60% of our workforce centers on R&D, guaranteeing nonstop innovation headway and investigating support.
Equipment Included
| Component | Specification | Function |
|---|---|---|
| Seed Cultivation Tanks | 500L-5,000L | Multi-stage strain preparation |
| Main Fermentation Reactors | 5,000L-100,000L | Core leucine production |
| Separation Systems | Continuous flow | Cell removal and clarification |
| Purification Equipment | Multi-column | Product refinement |
| Drying Systems | Spray/freeze drying | Final product processing |
| Control Systems | Automated PLC | Process monitoring and control |
| Quality Testing Lab | Complete analytical suite | Product verification |

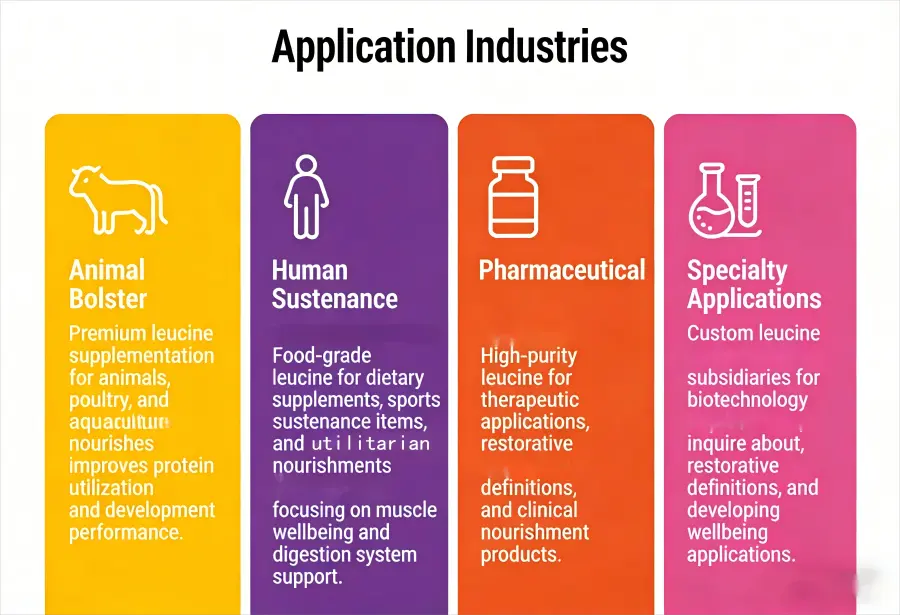
Application Industries
Animal Bolster: Premium leucine supplementation for animals, poultry, and aquaculture nourishes improves protein utilization and development performance.
Human Sustenance: Food-grade leucine for dietary supplements, sports sustenance items, and utilitarian nourishments focusing on muscle wellbeing and digestion system support.
Pharmaceutical: High-purity leucine for therapeutic applications, restorative definitions, and clinical nourishment products.
Specialty Applications: Custom leucine subsidiaries for biotechnology inquire about, restorative definitions, and developing wellbeing applications.
Certificate
- cGMP (Current Good Manufacturing Practice)
- FSSC22000 (Food Safety System Certification)
- ISO9001 Quality Management
- HACCP Food Safety
- HALAL and KOSHER Certifications
- Organic Production Standards

Turnkey Service
Project Planning & Design
Complete possibility thinks about, generation line format, and gear determination based on your capacity prerequisites and advertise positioning.
Installation & Commissioning
Full gear establishment, framework integration, and execution approval with ensured startup timelines.
Training & Support
Comprehensive administrator preparing, support conventions, and continuous specialized help to guarantee ideal generation performance.
Process Optimization
Continuous enhancement administrations counting abdicate upgrade, fetched diminishment procedures, and item quality upgrades.

FAQ
Q: What leucine purity levels can your technology achieve?
A: Our technology produces feed-grade (≥98%), food-grade (≥99%), and pharmaceutical-grade (≥99.5%) leucine depending on downstream purification intensity.
Q: How long does technology transfer take?
A: Complete technology transfer typically requires 6-12 months including equipment installation, commissioning, training, and production optimization.
Q: What raw materials does the process require?
A: Primary inputs include glucose/corn syrup, corn steep liquor, ammonia, and standard inorganic nutrients. All materials are readily available globally.
Q: Can existing fermentation facilities be adapted?
A: Yes, many existing facilities can be retrofitted with our technology. We provide detailed feasibility assessments and modification plans.
Q: What ongoing support do you provide?
A: Comprehensive support includes remote monitoring, troubleshooting, process optimization, strain maintenance, and equipment upgrades.
Contact & Consultation
Ready to implement advanced Leucine Production Technology? Contact our engineering team at producttech@asianbios.com for customized solutions and technical consultations.
Send Inquiry











